PRODUCT INFORMATION
Selectivity Profiling Assays
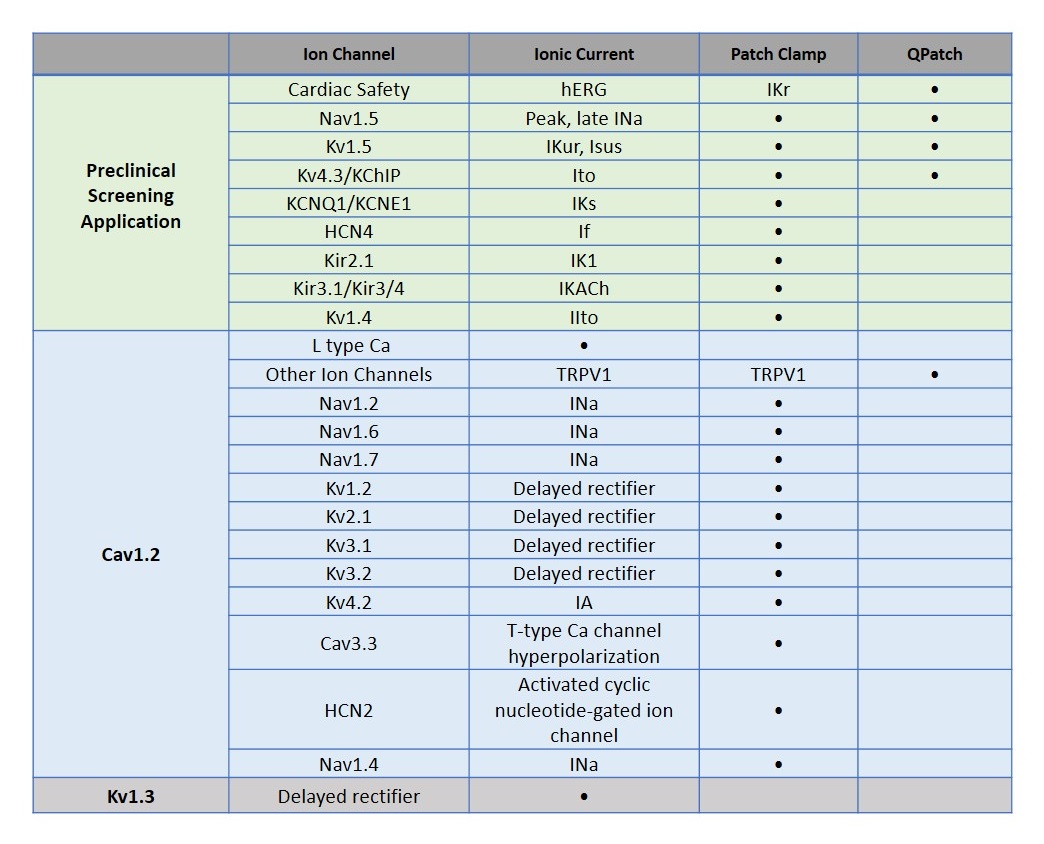
Inhibition of hERG alone is not specific enough to accurately predict either delayed repolarization or clinical proarrhythmia. The cardiac action potential results from a fine balance of activity between inward and outward ionic currents. Dysregulation of most ionic currents can lead to cardiac arrhythmias and/or mechanical dysfunction. In the context of the Comprehensive In vitro Proarrhythmia assay (CiPA) initiative, it becomes important to screen for an expanded panel of ion channels. Aurora offers multiple ion channel assays to better predict the proarrhythmic liability of your test compounds.
| NEW… Aurora now offers electrophysiological assays in human induced pluripotent stem cells-derived cardiomyocytes for characterization and screening of ion channel targets. Services are offered with high time resolution and sensitivity required for precise and direct characterization of ion channel properties. |
Ion Channel Profiling for Drug Discovery
Stable cell lines expressing various ion channels or transient expression of ion channels are used for Aurora’s electrophysiological assays. The electrophysiology gold standard, manual patch-clamp, provides you with the best quality data to make decisions on advancing molecules to the next phase of drug development. Isolated cardiac myocytes from various species are also available for patch-clamp studies.
Xenopus oocyte as a Screening Model
Xenopus oocytes are widely used as a cost-effective expression system for the functional characterization of ion channels. They are large cells easy to obtain, mechanically and electrically stable, and with low expression of endogenous ion channels. Due to the efficient translation of injected cRNA of interest, large-amplitude ionic current can be easily recorded using a two-microelectrode whole-cell voltage-clamp approach. This makes it a method of choice not only for screening test compounds to determine their relative efficacies against specific types of ion channels but also for determining the functional effects of mutations that cause human diseases.
Multiple ion channels are available on demand.
Reliable, Reproducible Results
The robustness of Aurora’s ion channel screening assays is shown in the figures below. No matter the stage of drug discovery, obtaining quick and accurate results is key to any research assay.
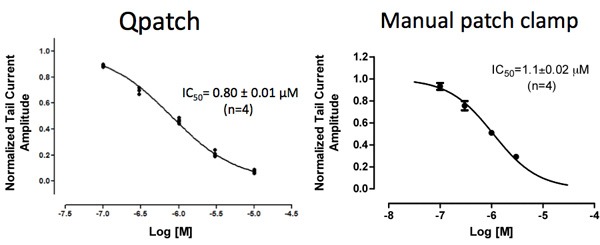
Figure 1: IC50 of flecainide for IhERG in CHO cells expressing hERG channels (Qpatch) and in HEK cells expressing hERG channels (manual patch clamp).
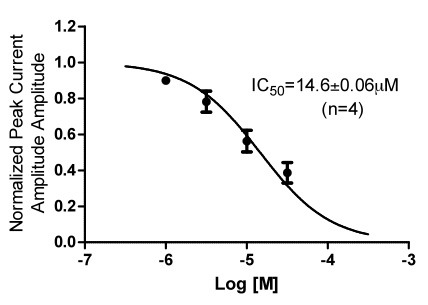
Figure 2: IKv4.2 block by flecainide in HEK cells expressing Kv4.2 channels by manual patch Clamp.
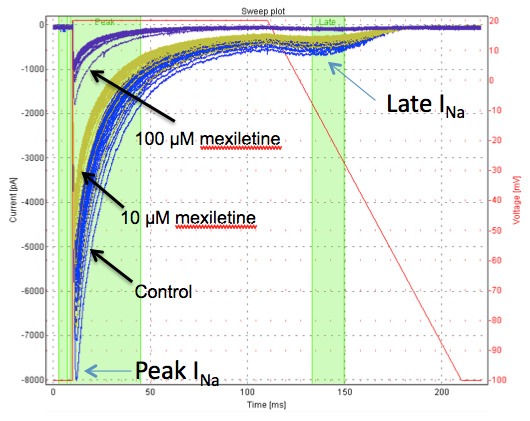
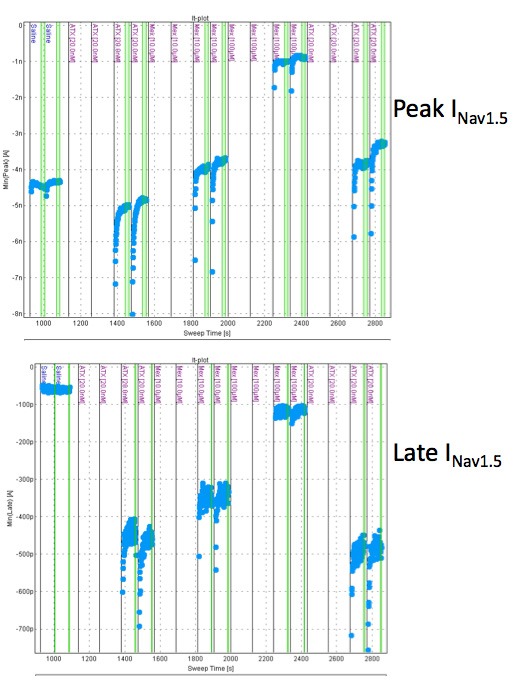
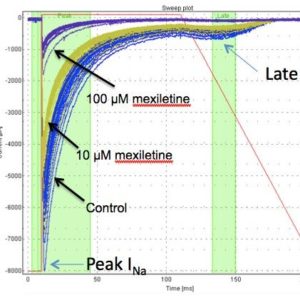
Figure 3: The effects of mexiletine on peak and late INav1.5 measured by Qpatch (in the presence of the sea anemone toxin ATXII)
Scope of Screening Services
Typical test compound screening services are completed as follows, depending on research requirements. Raw data can be sent (without analysis) upon request.
| Full Characterization | |
|---|---|
| 1 | a. Full DR curve with 5-7 points, each averaged from at least 3 independent estimations, or; b. Results are fitted to the Hill equation for accurate estimation of IC50. If the coefficient of variability (SD/mean) exceeds 25% at IC50, up to 6 independent estimates are provided for the same price. |
| 2 | Effect of the compound (at ~IC50) on I-V and G-V curves, including fit to Boltzmann equation and estimate of any shifts in G-V curve. |
| 3 | Effect of the compound on the kinetics of voltage-dependent inactivation of the channel at 3 voltages. |
| Partial Characterization | |
| 1 | DR curve with 4-5 points, each averaged from at least 3 independent estimations. |
| 2 | Effect of the compound (at IC50) on the I-V curve. |
| Preliminary Characterization | |
| 1 | a. Compound tested at a single dose in duplicate (n=2) at one voltage, or; b. Compound tested at two different doses at 3 voltages each (n=3). |
| Additional Characterization | |
| 1 | Effect of the compound on steady-state inactivation curve. |
| 2 | For N-type Ca2+ channels, additional estimates of the effect of the compound on channel regulation by G proteins, including estimation of the change in the extent of voltage-dependent facilitation and/or inhibition by a G protein-coupled receptor. |


