PRODUCT INFORMATION
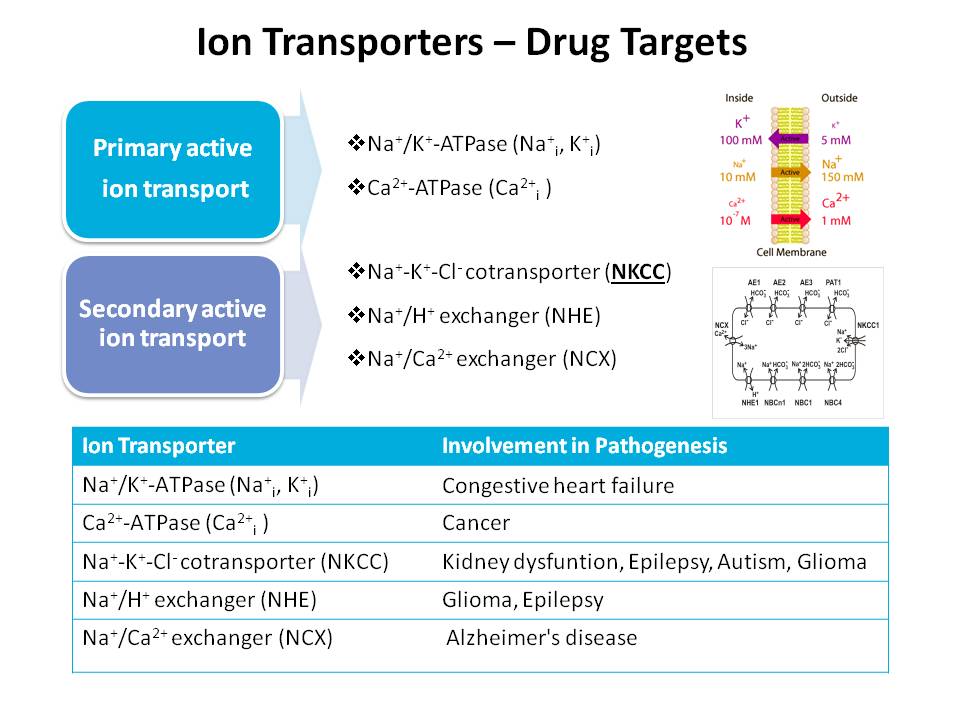
Transporters are key determinants of drug ADME properties. There are two classes of transporters. Many drugs have been proven to be substrates or inhibitors of ion transporters and would affect drug absorption, distribution, excretion, and interaction to different degrees. The importance of ion transporters in drug discovery has been growing increasingly, which would make a significant impaction on drug safety and efficiency. Several examples are listed below which show the involvement of the transporters in pathogenesis.
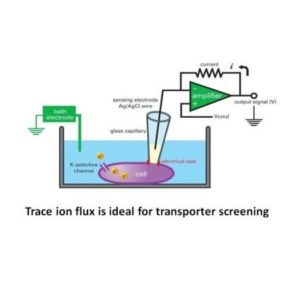
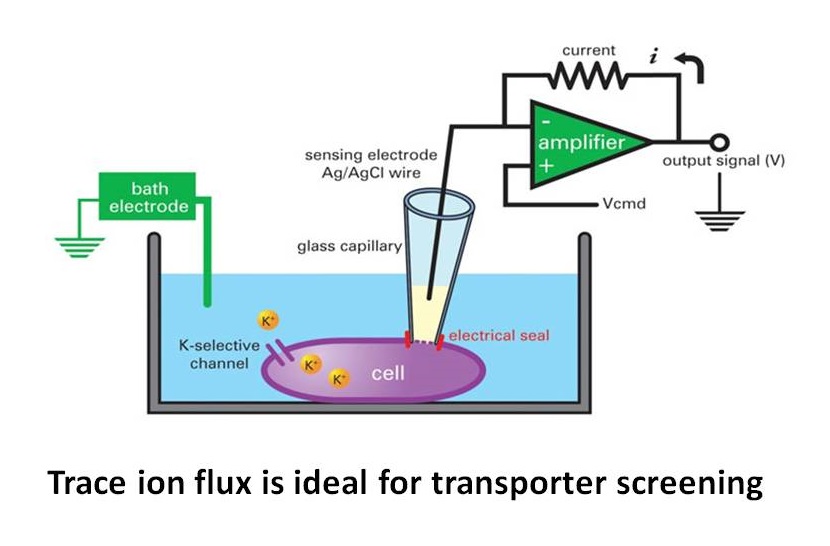
Trace ion flux is ideal for transporter screening
Transporters lack voltage sensors and cannot be as easily manipulated with a voltage clamp. Many co-transporters are electrically neutral and do not generate current. Considering labeling technologies such as radioactive or fluorescence-based assays there is a limited sensitivity and limited molecule turnover per seconds. Electrophysiological technologies also have little to no measurable current as ion flux through transporters is usually too slow and small. Trace Ion Flux is one of the best methods capable of obtaining high throughput activity measurements for electro-neutral ion transporters.
Non-radioactive Rubidium Flux Assay in Ion Transporters Research
Small and sometimes electroneutral ion fluxes can limit the application of conventional EP methods to ion transporter study. Aurora’s ICR and assay services is an optimal technology for both functional and pharmacological transporter study.
Ion transporters are membrane proteins that facilitate the passage of ions against their concentration gradients. They are significant drug targets that have broad implications in drug discovery and development. The ion flux of transporters is much lower than what would be observed in the case of ion channels and in some cases is non-existent. This makes study by conventional means quite difficult. Over the past few years, we have worked diligently to refine our assays to be able to screen compound libraries and perform structure-function assessments of ion transporters.
One example is the high throughput screening of NKCC1 Modulators (Roche) – NKCC1 transporters produce an electroneutral flux across cell membranes, making study by patch-clamp impractical. A study published in conjunction with Roche details the ICR as a highly effective tool for high throughput screening of NKCC1 channels. As a result of this study, several hits were obtained from a focused library of 1450 compounds.
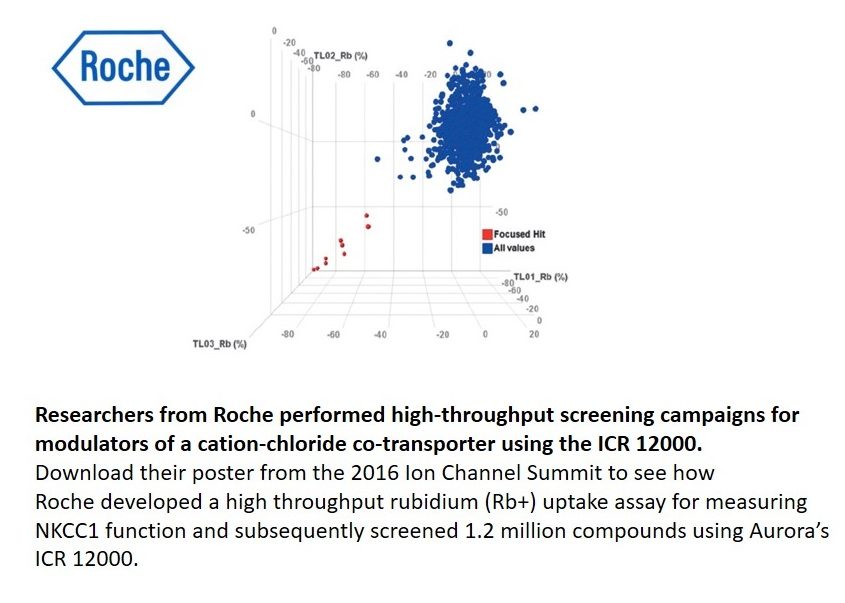
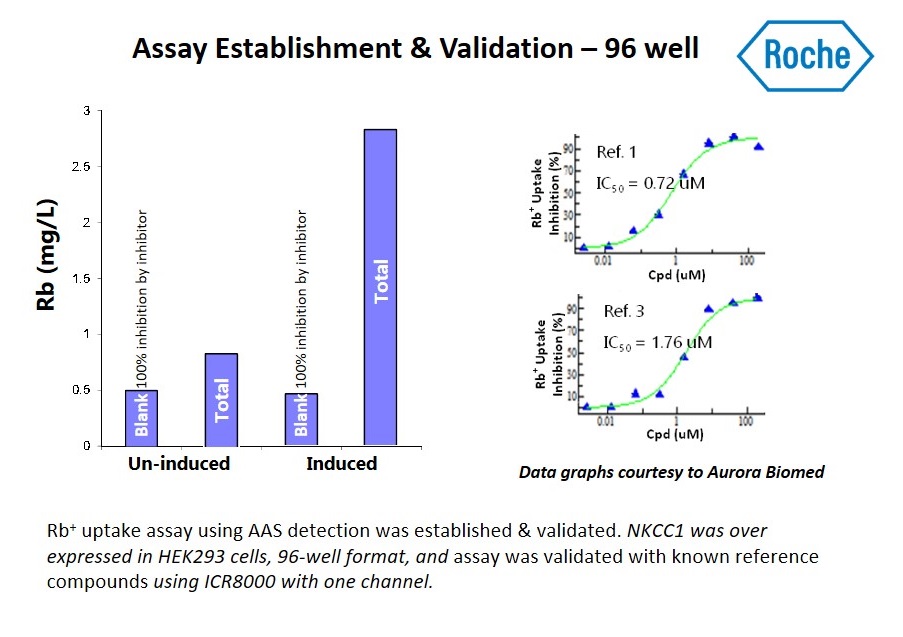
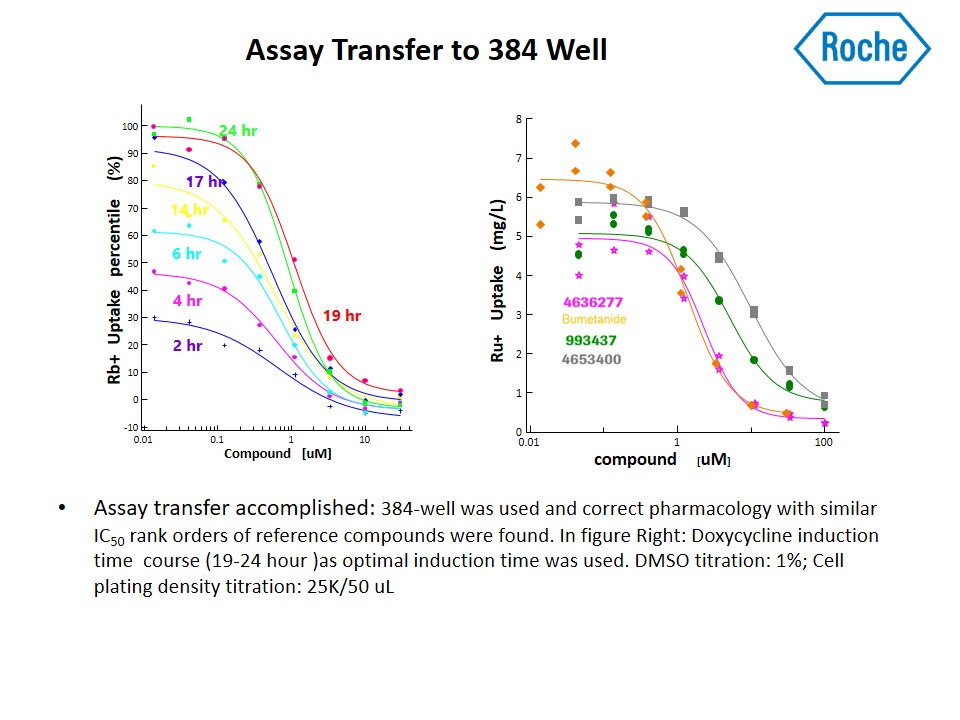
Researchers from Roche performed high-throughput screening campaigns for modulators of a cation-chloride co-transporter using the ICR 12000.
Download their poster from the 2016 Ion Channel Summit to see how Roche developed a high throughput rubidium (Rb+) uptake assay for measuring NKCC1 function and subsequently screened 1.2 million compounds using Aurora’s ICR 12000.
Recently Published Papers (2020)
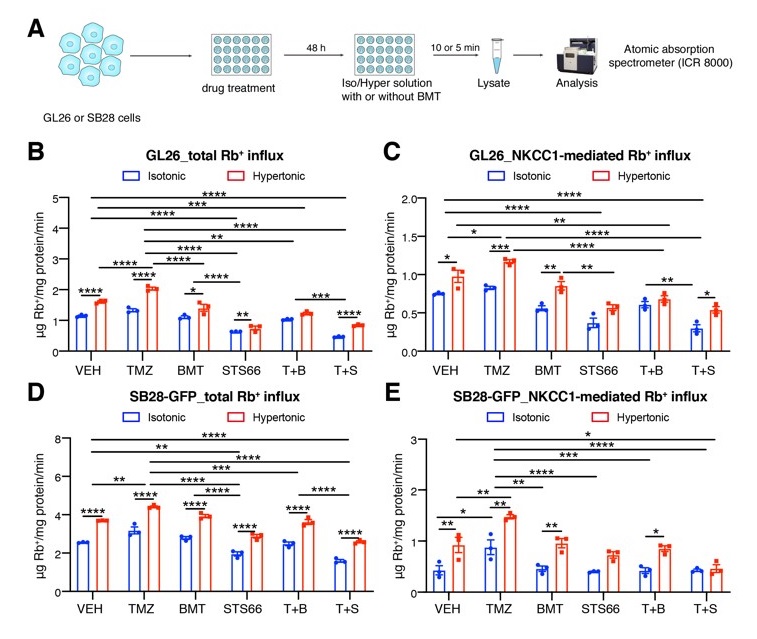
More recently Aurora’s ICR technology has been used for high throughput screening of NKCC1 modulators and the finding on the Role of NKCC1 activity in glioma K+ homeostasis and cell growth has been published by a research group at the University of Pittsburgh. Na+-K+-2Cl- cotransporter isoform 1 (NKCC1) is an important transporter that regulates intracellular K+ and Cl- homeostasis and cell volume. Researchers at the U of Pittsburgh investigated the role of NKCC1 in regulating glioma K+ influx and proliferation in response to apoptosis-inducing by temozolomide (TMZ) chemotherapeutic drug. The efficacy of STS66, a new BMT-derivative NKCC1 inhibitor in blocking NKCC1 activity was compared with bumetanide (BMT), a well-established NKCC1 inhibitor. NKCC1 activity in cultured mouse GL26 and SB28-GFP glioma cells was measured by Rb+ (K+) influx using Aurora’s ion channel reader 8000 (ICR 8000).
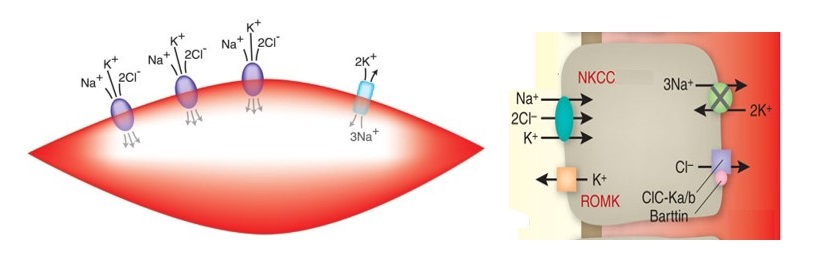
Importance of NKCC
The Na-K-Cl cotransporter known as (NKCC) is a protein that helps the secondary active transport of sodium (1Na+), potassium (1K +), and chloride (2Cl –) ions into cells. NKCC mainly is present in tissues including brain and kidney and is considered as an important drug target in hypertension, epilepsy, and neonatal seizures. In humans there are two isoforms of NKCC known as NKCC1 and NKCC2, encoded by two different genes.