Things to keep in mind with rapid antigen tests:
- Faintness, or how dark the line is does not change the result. If the line is there then it should be read.
- Results are only valid for the time on the package. Typically this time period is 15-30 minutes. Results after the time on the box should NOT be read.
- The C stands for Control, the T stands for Test.
- If you have questions about your diagnoses contact your doctor or healthcare provider.
The COVID-19 Antigen Rapid Test is a rapid membrane-based lateral flow immunoassay for the qualitative detection of SARS-CoV-2 antigens in human nasopharyngeal swab specimens. The use of a COVID-19 Antigen Rapid Test allows you to detect the presence of SARS-CoV-2 virus antigens in our body and to take initial precautionary isolation measures in order to reduce further spread of the virus. These tests are particularly useful for identifying a person who is at or near peak infection. So the question then is, how do I interpret the antigen rapid test result?
COVID-19 affects different people in different ways. If you are felling the symptoms use a rapid antigen test at home.
COVID-19 Antigen Rapid Test Cassette Structure
The COVID-19 Antigen test cassette is coated with two lines (control line and the test line). If the specimen contains SARS-CoV-2 antigens, they will bind to the SARS-CoV-2 antibodies coated on the (T) Test line region and generate a colored line on the test strip, indicating a positive result. If the antigens are not present in the specimen, no colored line will appear in the (T) Test line region, indicating a negative result. As a procedural control, a colored line will always appear in the (C) Control line region, indicating that the test procedure has been performed properly and that test components have operated as intended.
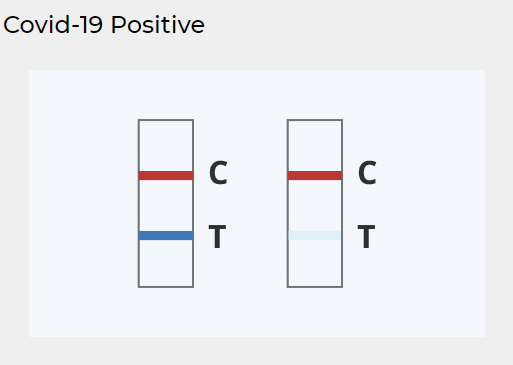
Look at the images below
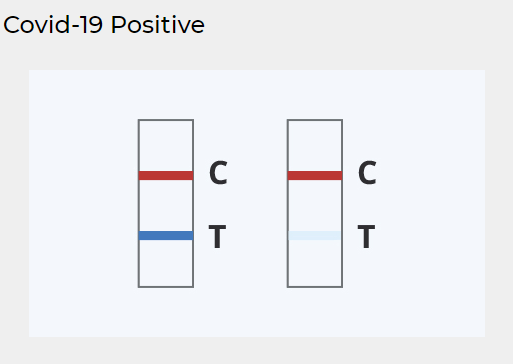
Keep reading for more information about interpreting COVID-19 results
Valid Antigen Rapid Test Results
POSITIVE RESULT:
Two colored lines appear
A colored line should always appear in the Control (C) region and another line should be in the Test (T) region. A positive result indicates detection of SARS-CoV-2 antigens in the sample.
A positive antigen test result is considered accurate when instructions are carefully followed, but there’s an increased chance of false-negative results — meaning it’s possible to be infected with the virus but have a negative result.
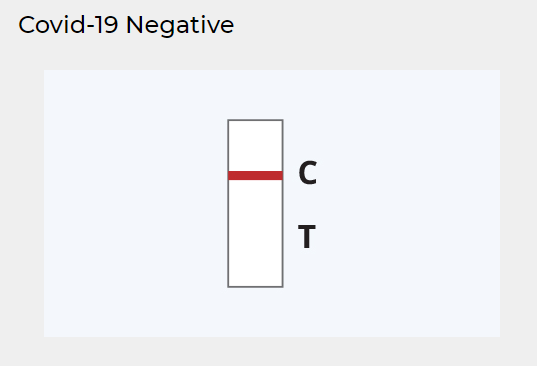
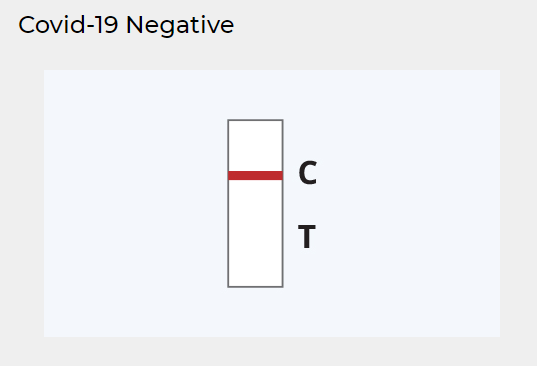
NEGATIVE RESULT:
A colored line appears in the Control (C) region.
No line appears in the Test (T) region.
As with the positive result, a colored line should always appear in the Control (C) region for the test to the valid. A negative result indicates that the detection threshold of SARS-CoV-2 antigens in the sample has not been reached. There is still the chance of an infection if the concentration of antigens in the sample is too low to be detected by the rapid antigen test.
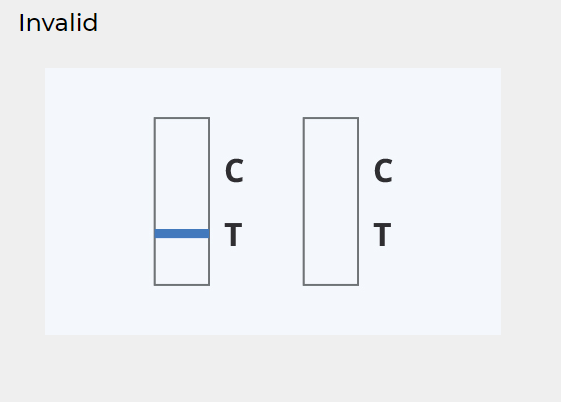
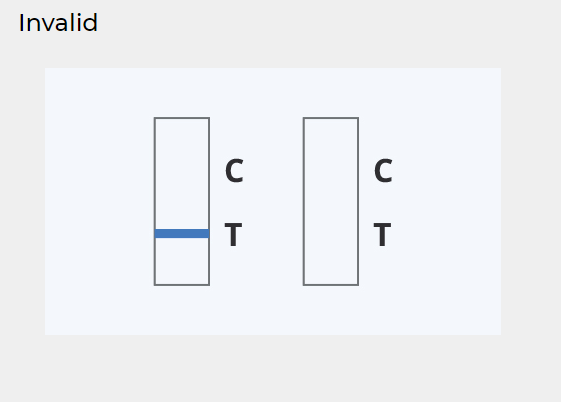
INVALID RESULT:
The control line does not appear.
Insufficient diluent volume or incorrect procedural techniques are the most likely reasons for the absence of the control line. Review the procedure and repeat the test with a new device.
Antigen tests are relatively inexpensive, and most can be used at the point of care. Most of the currently authorized tests return results in approximately 15 minutes. Antigen tests for SARS-CoV-2 are generally less sensitive than real-time reverse transcription-polymerase chain reaction (RT-PCR) and other nucleic acid amplification tests (NAATs) for detecting the presence of viral nucleic acid. However, RT-PCR can detect levels of viral nucleic acid that cannot be cultured, suggesting that the presence of viral nucleic acid does not always indicate contagiousness.
*Please interpret the result of rapid antigen and antibody tests within 15-30 minutes. The result is not valid if you check again in a few hours.