
PRODUCT INFORMATION
Third deadly CoV outbreak
In less than 20 years, three deadly international CoV outbreaks occurred. The first one was the Severe Acute Respiratory Syndrome (SARS) CoV-1 outbreak that started in 2003. The second one was the Middle East Respiratory Syndrome (MERS) CoV occurred in 2012 and currently, we are experiencing the SARS-CoV-2 pandemic starting from 2019.

Evidence for ion channel activity in CoV proteins and potential for modulation as a therapeutic approach
COVID-19 Drug & Vaccine Development
From early 2020, several hundred pharmaceutical and biotechnology companies, university research groups, and health organizations started developing therapeutic candidates for COVID‑19 disease. Considerable attention has been made to spike proteins on the surface of the virus. While the role of other proteins with ion channel activity is less clear. Sequencing of the virus genome revealed the presence of several envelope E proteins and putative ion channel genes. Since then numerous academic groups have begun to study and target COVID-19 viral ion channels (viroporins). Interestingly it has been proposed that Sars-Cov2 ion channels might have contributions to inflammation and severity of COVID‑19 disease. It has been also shown that the 2-E protein not only exists on the surface of the virus but also is overexpressed on the membranes of host cells and forms cation channels, which eventually lead to membrane rupture and tissue damage. In vitro and in vivo experiments revealed that some channel inhibitors have potent anti-SARS-CoV-2 activity and can protect the tissue against the 2-E-induced damage.
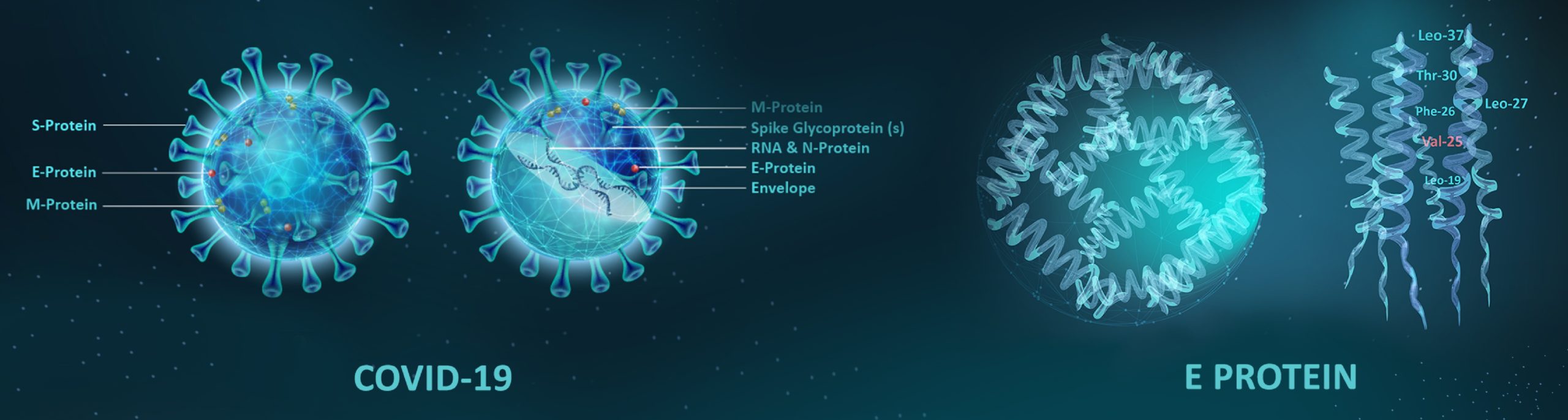
Coronavirus (SARS-CoV-2) is enclosed in an envelope bilayer (Left) having membrane (M), envelope (E) & spike (S) structural proteins. Viroporins, integral viral membrane ion channel proteins, also known as E proteins (Right) and have shown ion conductance properties in membranes and are proposed as therapeutic targets.
Altogether the findings so far support that CoVs also code for their own ion channels. ORF3a & E are potential ion channels for SARS-CoV/SARS-CoV-2 that have been proposed as candidates of drug targets. According to evidence, 2-E is a promising drug target against SARS-CoV-2 and forms a type of pH-sensitive cation channel that functions as ion channels in the viral membranes, as well as host cells. SARS-CoV E protein has a lumenal N-terminus & a cytoplasmically oriented C-terminus, a champagne flute shape structure, with the wider opening facing the cytoplasmic side. At the moment good channel inhibitors are lacking and their availability will help to reveal the specific role these channels play in the life cycle of these viruses as well as the development of drug candidates for targeting them. Aurora’s series of automated Ion Channel Readers (ICRs) obtain high throughput activity measurements for ion channels by measuring the absolute concentrations of cytosolic and extracellular ions. This technique is independent of and complementary to methods that rely on voltage manipulation and could be used for screening of compounds used as potential therapeutics. Automated ICRs (Ion Channel Readers) combines the precision and sensitivity of atomic absorption spectroscopy (AAS) with patented microsampling processes and liquid handling technologies to create a high-throughput screening solution for ion channel researchers that fills gaps which automated patch-clamp cannot. By detecting ion channel activity using atomic absorption spectroscopy and flux assays, the ICR series of automated Ion Channel Readers can analyze ion channels regardless of electrogenicity. ICR technology can be used with both voltage-gated (e.g., hERG, Kv1.1, Nav1.5) and ligand-gated (e.g., KATP, nAChR) channels, as well as with ion pumps and transporters (e.g., Na+/K+-ATPase), allowing researchers to accelerate drug development for the treatment and prevention of diseases associated with Coronavirus disease.
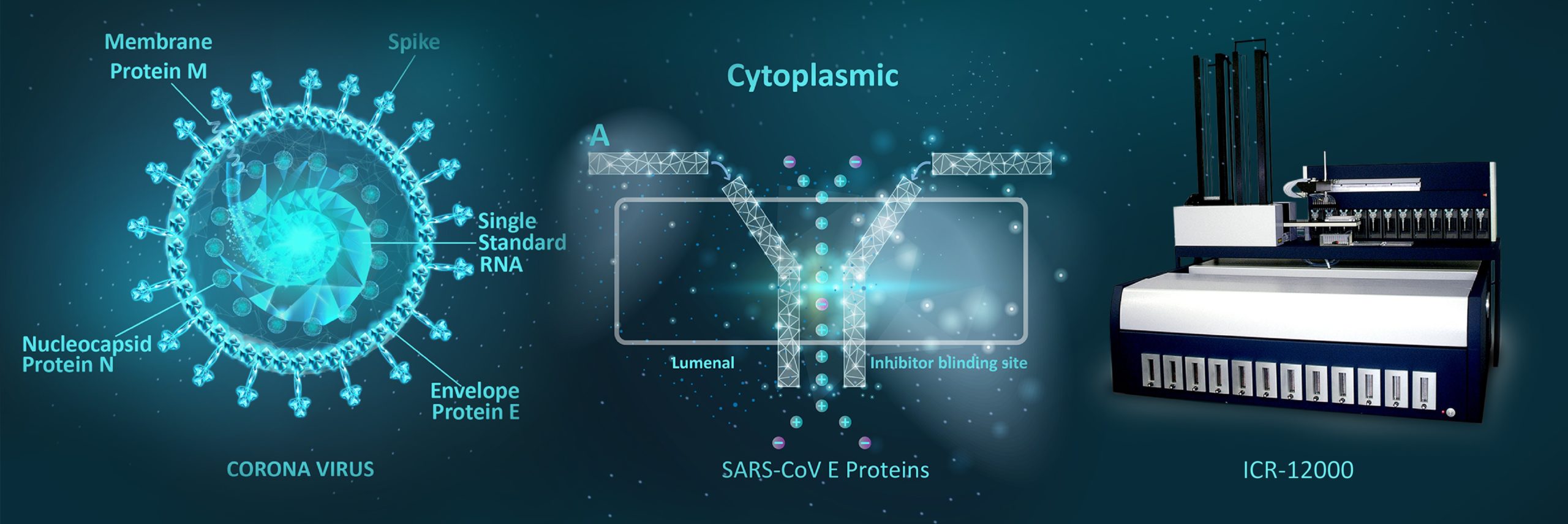
Application of Ion Channel Technology (ICRs & Flux Assay) in Drug Discovery & Development of Coronavirus Disease 2019 (COVID-19) – Targeting Ion Channels
Targeting Virus/Host Ion Channels
COVID‑19 drug and vaccine development is the procedure of developing preventative therapeutic drugs and vaccines that would alleviate the severity of COVID‑19 disease. As it is acknowledged Ion channels are one of the major drug targets that account for 13 % of FDA drugs. In the case of the Covid-19 outbreak, similar to other viral infections there are two therapeutic methods for targeting ion channels:
1) Inhibition of host ion channels with multiple ion channel modulators that have been repeatedly shown to affect virion entry and endosomal fusion.
2) Identification of viral ion channels that allows screening for novel virus-specific channel modulators
Potential antiviral targets: Since the initial characterization of influenza M2, viral proteins which form ion channels themselves (viroporins) or which can modulate host cell ion channel function (e.g. HIV-1 Vpu), have been reported in a variety of virus species, and repeatedly proposed as potential antiviral drug targets. For example, influenza M2 was shown as a target for anti-influenza A drug discovery, and M2 blockers (Amantadine & Rimantadine) were developed. And in case of the Hepatitis C virus, p7 ion channel protein was shown as a potential antiviral target.
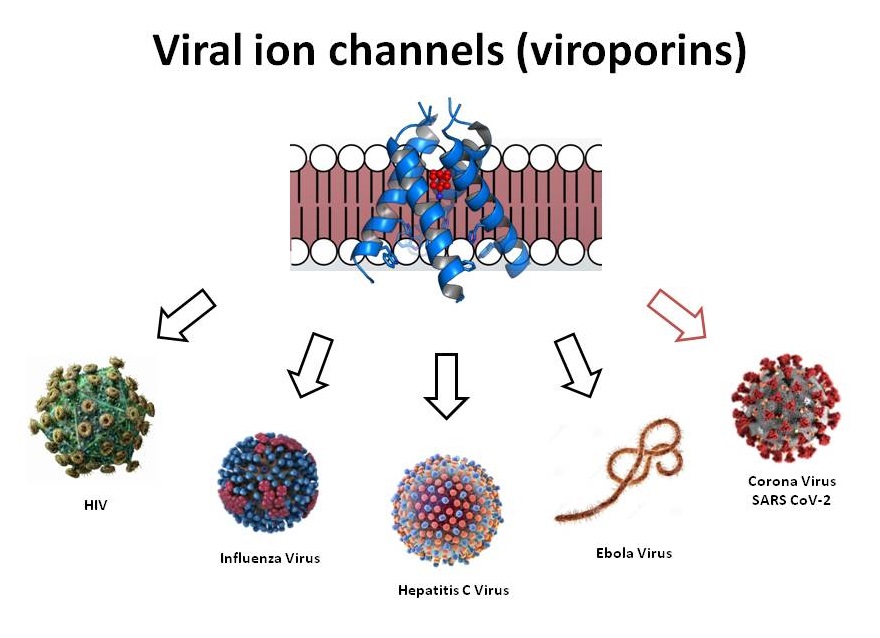
Viral ion channels forming proteins (viroporins) also identified in Severe Acute Respiratory Syndrome (SARS)-CoV-2 and exhibit low degree of homology with ion channels of prokaryotic or eukaryotic origin. Although their transmembrane (TM) regions do bear some resemblance to the corresponding regions of ion channels of higher organisms. Considering the coronavirus family, ion channels mainly operate at several steps in the viral life cycle including regulation of replication compartment, viroplasm formation, virion budding, and viral infection. Lack of homology between CoV viroporins and human ion channels provides the potential for selective modulation with small molecule or biologic therapies. Aurora’s Ion channel reader (ICR technology) allows for the high throughput screening of novel channel modulators.
Electrophysiological characterization of E Proteins
Of all coronavirus proteins, E is the least understood. Functionally the E protein is involved in viral assembly, release, and pathogenesis. Small size and overall hydrophobicity prompted suggestions that coronavirus E proteins as viroporins. Synthetic SARS-CoV E, or E proteins from mouse hepatitis virus (MHV)/infectious bronchitis virus (IBV) was reconstituted into artificial bilayers. Reports demonstrated that these proteins cause fluctuating currents and indistinct gating events. Attenuated viruses lacking the E protein have even been suggested to serve as vaccine candidates. E proteins from several coronaviruses including SARS-CoV-1, MERS coronavirus, human coronavirus, mouse hepatitis virus, and infectious bronchitis virus were shown to possess cation-selective channel activity that may be blocked by compounds like Hexamethylene amiloride. Aurora’s non-radioactive assay as a screening tool of membrane protein modulators is well-documented in scientific literature and has been widely used for studying the potassium channel family. Rubidium is the most commonly used tracer ion to study potassium channels because of its similar physical properties to K, little natural presence in physiological systems, and ease to detect by AAS. The principle of the non-radioactive Rb assay can be easily applied to other membrane protein targets as well.


